| Professor | Hozumi MOTOHASHI |

Redox metabolism underlying gene expression regulation
The movement of electrons—that is, redox (oxidation–reduction) reactions—constitutes one of the most fundamental processes of life. Redox reactions not only drive energy metabolism, which sustains all biological activities, but also underpin the mechanisms that maintain homeostasis in the oxygen-rich environment of present-day Earth. Disruption of redox homeostasis imposes stress on living systems and triggers adaptive responses.
In our laboratory, recognizing that the extension of healthy life expectancy is one of the most pressing challenges in medicine in this rapidly aging society, we seek to understand the biological stress response mechanisms that enable organisms to adapt to environmental changes and sustain life.
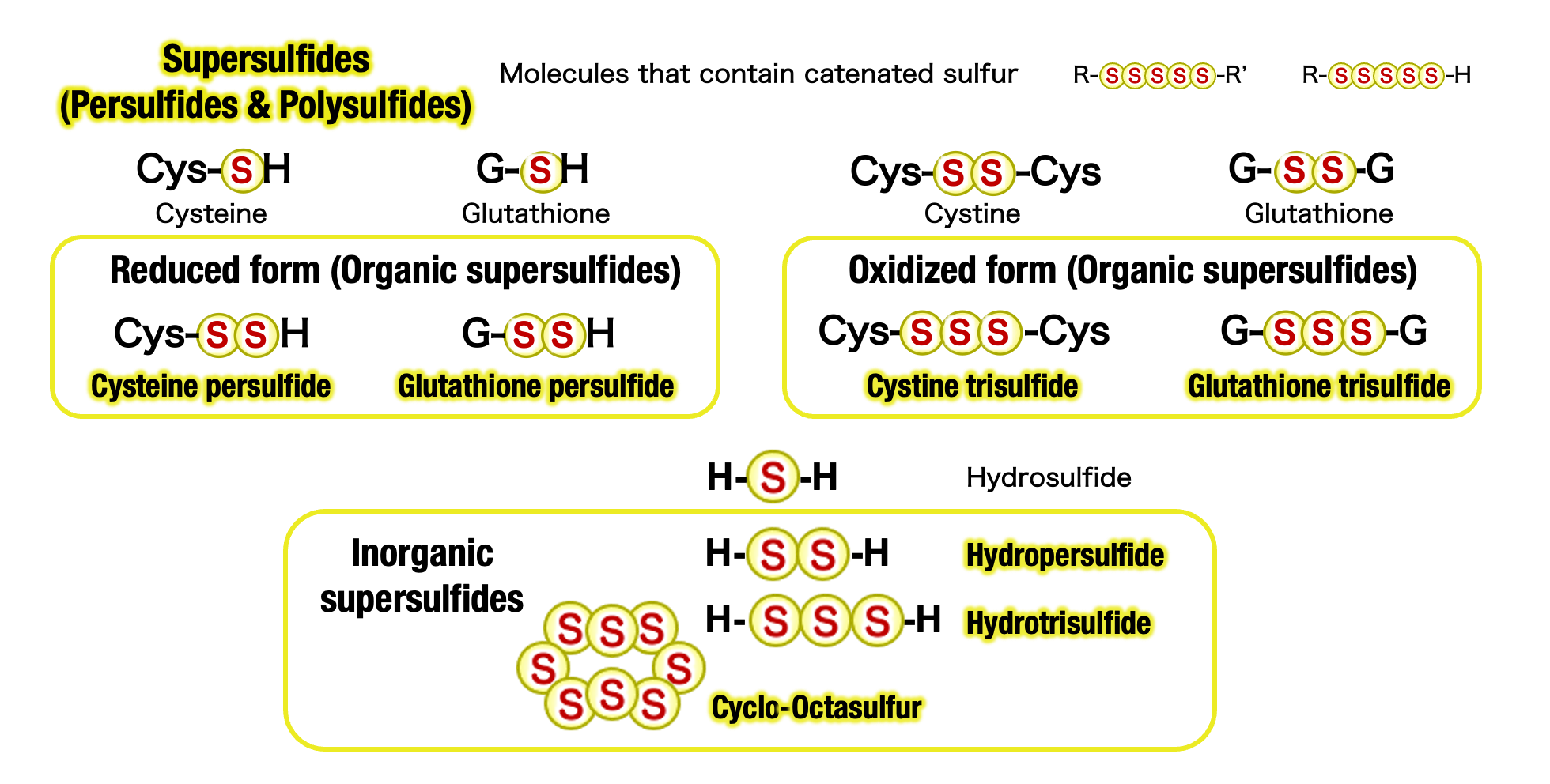
Supersulfides as a New Entity of Biomolecules
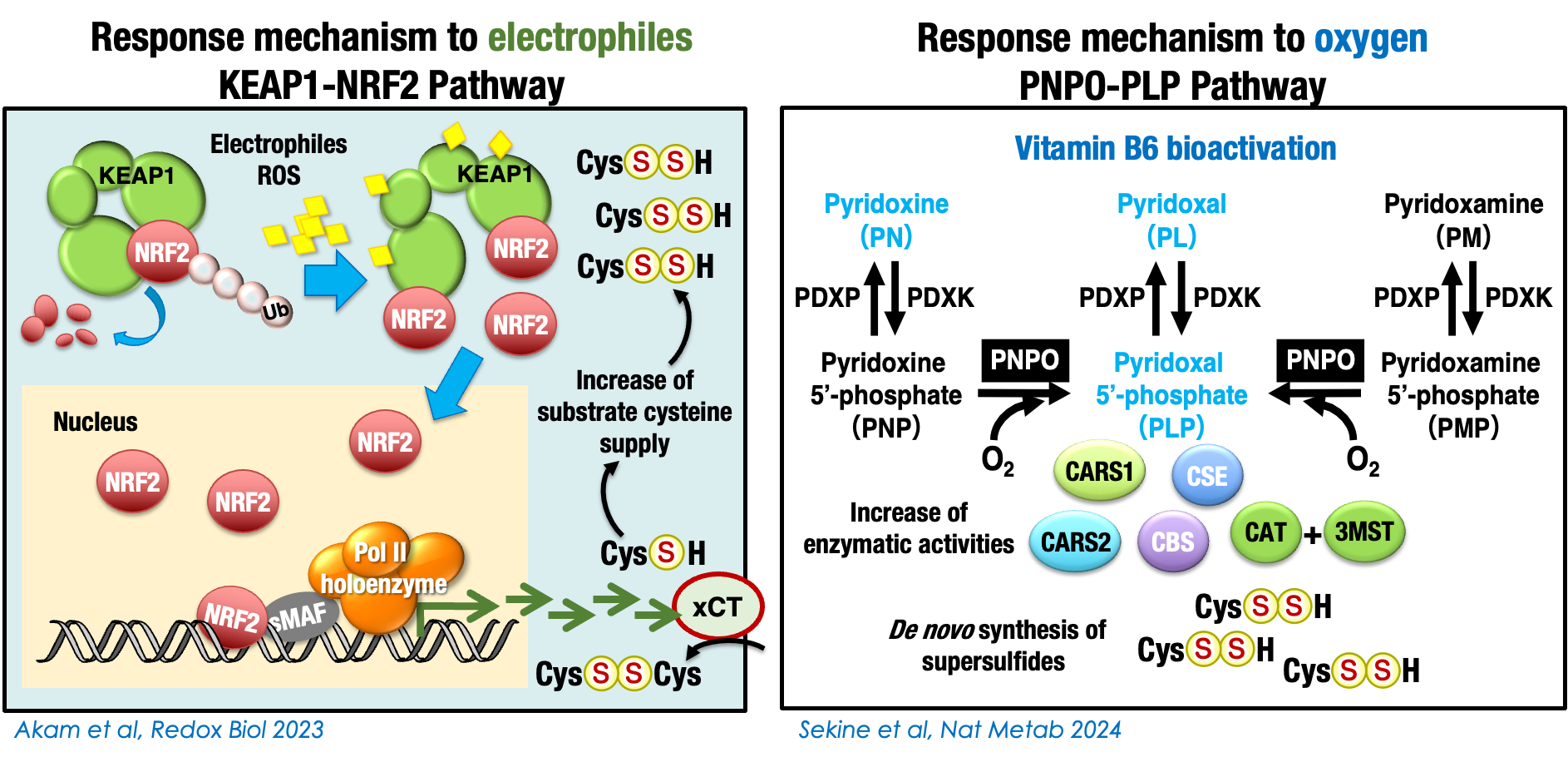
Two regulatory Pathways for Synthesis of Cysteine Persulfide as a Primary Supersulfide
Research Topics
・Physiological and pathological roles of supersulfides and regulatory mechanisms of their synthesis.
・Chalcogen metabolism for stress response to oxygen.
・Biological and therapeutic significance of reductive stress in intractable cancers.
・Pathological basis of inflammatory bowel diseases emerging from metabolic rewiring.
Selected Publications
1. Sung E, Murakami S, Morita M, Ida T, Akaike T, Motohashi H. Mitochondria regulate the cell fate decisions of megakaryocyte-erythroid progenitors. Stem Cell Rep. 2025 Nov 20:102720. doi: 10.1016/j.stemcr.2025.102720.
2. Sekine H, Akaike T, Motohashi H. Oxygen needs sulfur, sulfur needs oxygen: a relationship of interdependence. EMBO J. 2025 Jun;44(12):3307-3326. doi: 10.1038/s44318-025-00464-7.
3. Sekine H, Takeda H, Takeda N, Kishino A, Anzawa H, Isagawa T, Ohta N, Murakami S, Iwaki H, Kato N, Kimura S, Liu Z, Kato K, Katsuoka F, Yamamoto M, Miura F, Ito T, Takahashi M, Izumi Y, Fujita H, Yamagata H, Bamba T, Akaike T, Suzuki N, Kinoshita K, Motohashi H. PNPO-PLP axis senses prolonged hypoxia in macrophages by regulating lysosomal activity. Nat Metab. 2024 Jun;6(6):1108-1127. doi: 10.1038/s42255-024-01053-4.
4. Takeda H, Murakami S, Liu Z, Sawa T, Takahashi M, Izumi Y, Bamba T, Sato H, Akaike T, Sekine H, Motohashi H. Sulfur metabolic response in macrophage limits excessive inflammatory response by creating a negative feedback loop. Redox Biol. 2023 Jul 29;65:102834. doi: 10.1016/j.redox.2023.102834.
5. Alam MM, Kishino A, Sung E, Sekine H, Abe T, Murakami S, Akaike T, Motohashi H. Contribution of NRF2 to Sulfur Metabolism and Mitochondrial Activity. Redox Biol. 60, 102624, 2023. doi: 10.1016/j.redox.2023.102624.
Research Interests
Sulfur metabolism, KEAP1-NRF2 pathway, PNPO-PLP pathway, Oxiudative stress, Reductive stress
